 
It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. 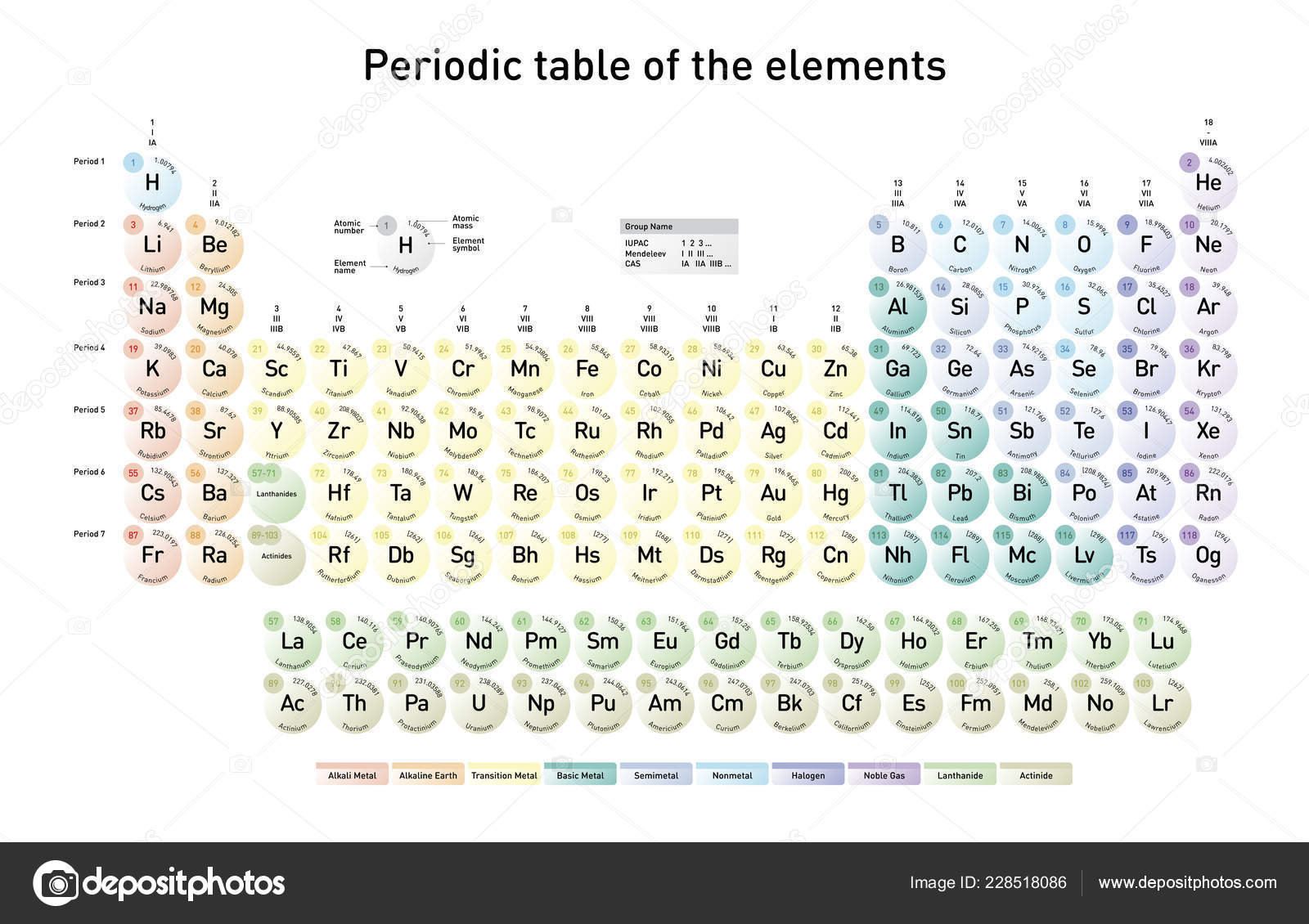
Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Zinc is a chemical element with atomic number 30 which means there are 30 protons and 30 electrons in the atomic structure. K) 30.2 Thermal Conductivity 116 Specific Heat 0.39 Heat of Fusion 7.322 Heat of Vaporization 115.3 Atomic Number of Zinc.You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Zinc – Properties Element Zinc Atomic Number 30 Symbol Zn Element Category Transition Metal Phase at STP Solid Atomic Mass 65.409 Density at STP 7.14 Electron Configuration 3d10 4s2 Possible Oxidation States +2 Electron Affinity - Electronegativity 1.65 1st Ionization Energy 9.3941 Year of Discovery unknown Discoverer unknown Thermal properties Melting Point 419.53 Boiling Point 907 Thermal Expansion µm/(m Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Let me know in the comments, has this article helped you or not? Free Gift for you: Interactive Periodic Table I hope you have completely understood the arrangement of Periodic table. There are 4 blocks on the Periodic table which describes the position of valence electrons in the s, p, d or f orbitals. Later on, we saw that there are groups as well as periods on the Periodic table. Hence for grouping the elements according to the similar chemical properties, the elements of the Periodic table are arranged in the increasing order of their atomic number. These electrons are responsible for the chemical properties of an element. In the very beginning of this article, we have seen that the elements of periodic table are arranged in the increasing order of their atomic number.Īnd for a neutral atom, the number of protons are equal to the number of electrons. Similarly, elements which are in the p block have the valence electrons in p-orbitals.Įlements in d block have valence electrons in d-orbitals and elements of f block have valence electrons in f-orbitals. The elements which are in the s block have the valence electrons in s-orbitals. There are 4 blocks in the Periodic table: s block, p block, d block and f block. The elements of the Periodic table are also classified block wise. The first element having the least atomic number 1 (i.e hydrogen) is placed on the first cell of the Periodic table. So now you know that the elements are arranged according to the increasing order of their atomic number. Step by step arrangement (from 1 st to 118 th element) Hence the Periodic table is organized on the basis of Atomic number. In other words, Atomic number indicates the number of electrons, which are responsible for the chemical properties of elements. Hence to classify the elements on the basis of similarities in their chemical properties, they are arranged in the Periodic table on the basis of atomic number. The electrons present in the outermost orbit represent the chemical properties of the elements. So far in the above discussion, we have seen that the atomic number represents the number of electrons of an element. Number of protons (or atomic number) = Number of electrons Summary (Why is Periodic table arranged by atomic number?) Here you can see that the helium atom has 2 protons and the number of electrons are also 2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
